- Blog
- Hello neighbor alpha 1 vs alpha 2
- Maintenance checklist car
- Tree cad blocks free download
- Valence electrons in the periodic table
- Queen victoria family tree ppt hemophilia
- Free ant jailbreak vip server
- Genogram 3 generations
- Draco airplane coloring page
- Pny usb format tool windows
- Sakura dungeon installing patch breaks game
- Happy wheels full game free 2
- Foxit phantompdf 7-2 activation key free
- Hearts card game play it online
- Windows 8-1 professional product key 64 bit
- Exploit download trigon
- The ballad of jane doe vocal range
- Rage hook plugin 1180 -57 download
- Mother and baby animal matching
- Waifu sex simulator without vr headset
- Portable google chrome without installation
- Barnes and noble collection
- Winzip pro activation code list latest version
- Windows 7 update stuck internet explorer 11
- Avery labels 18163 template
- Moving checklist for new location
- Daily schedule template editable
- 3d maps with rpg maker vx ace
- Elvenar gems of knowledge tales
- Iei outdoor keypad
- What is the bonzi buddy voice
- Waifu Sex Simulator VR 2-3 torrent
- Archeage map of shipwrecks
- Live wallpaper pc anime
- Shadowrun 5e character sheet pdf
- Barney-s campfire sing along vhs vhs tape
- Autodesk inventor 2013 descargar
- Standard poster frame sizes
- Emcee send off wedding script
- Download full version of sql server 2012
- Direct3d overrider windows 10 download
- Best online webm to gif converter
- Loading gif aesthetic transparent
- Youtube music to mp3 download free
- Free small business software downloads
- Download putty and puttygen for windows 10
- Slime rancher multiplayer mod -
- Festo fluidsim 5 demo
- Free holiday border for word document
- Match baby animal names game
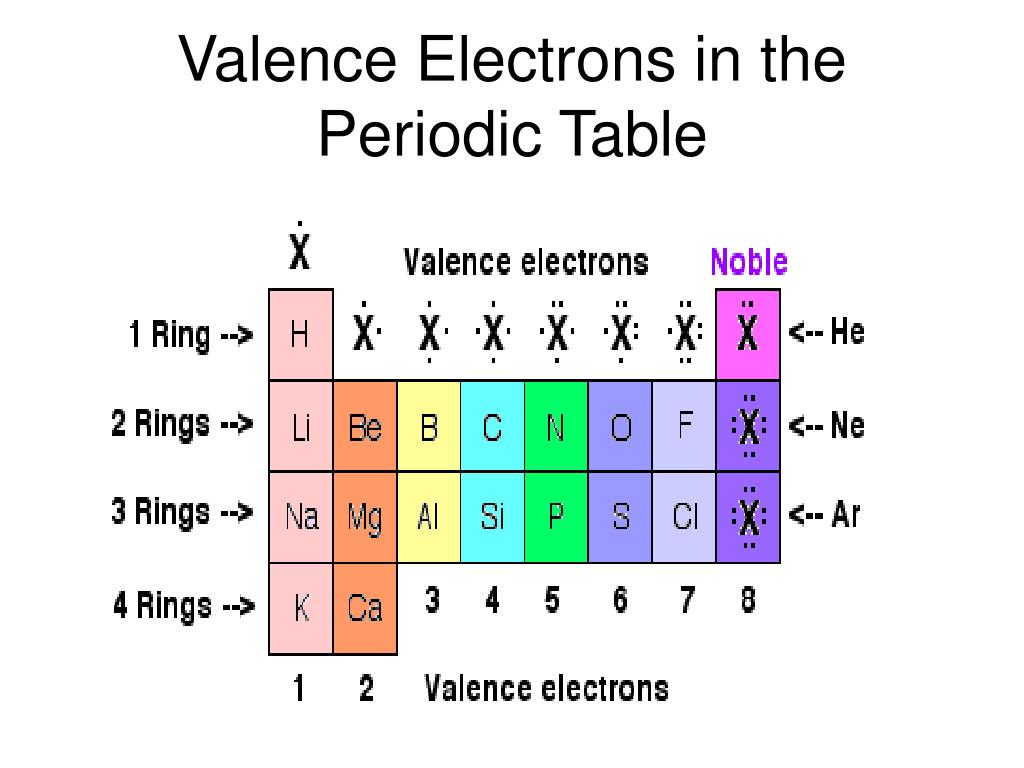
We know today that the observed properties of the elements are determined by their outermost electron cloud or shell, which has been described as the rule of octet rule. One of the most striking patterns reveals an octet arrangement, or that certain characteristics of the elements repeat every eight elements. Where there were elements missing, he left room for elements that were yet to be discovered. Mendeleyev probably wrote the properties of elements known at the time on placards and arranged them in similar groups to discover that they were repeating, or periodic, thus the name of the table today, the periodic table. atoms can combine in ratios to form compounds.all atoms of an individual element have the same properties including mass.that elements were made of small indivisible particles called atoms.Mendeleyev predicted the existence of previously unknown elements and paved the way for modern chemistry. The first periodic table that ordered the elements based on their atomic weight was developed by Dmitri Mendeleyev, a Russian chemist, in the 1860s. The last lessons concentrated on the chemical and physical aspects of biology. We have covered an extensive (but not exhaustive) review of the main disciplines of the sciences: scientific investigation, physics, chemistry, Earth science, and biology. The material will concentrate on empirical characteristics of the elements that are reflected by their valance electrons.

In this lesson, we will be reviewing the properties of the periodic table and the patterns of the elements displayed therein. Tools of the Trade: the Periodic Table Objective
- Blog
- Hello neighbor alpha 1 vs alpha 2
- Maintenance checklist car
- Tree cad blocks free download
- Valence electrons in the periodic table
- Queen victoria family tree ppt hemophilia
- Free ant jailbreak vip server
- Genogram 3 generations
- Draco airplane coloring page
- Pny usb format tool windows
- Sakura dungeon installing patch breaks game
- Happy wheels full game free 2
- Foxit phantompdf 7-2 activation key free
- Hearts card game play it online
- Windows 8-1 professional product key 64 bit
- Exploit download trigon
- The ballad of jane doe vocal range
- Rage hook plugin 1180 -57 download
- Mother and baby animal matching
- Waifu sex simulator without vr headset
- Portable google chrome without installation
- Barnes and noble collection
- Winzip pro activation code list latest version
- Windows 7 update stuck internet explorer 11
- Avery labels 18163 template
- Moving checklist for new location
- Daily schedule template editable
- 3d maps with rpg maker vx ace
- Elvenar gems of knowledge tales
- Iei outdoor keypad
- What is the bonzi buddy voice
- Waifu Sex Simulator VR 2-3 torrent
- Archeage map of shipwrecks
- Live wallpaper pc anime
- Shadowrun 5e character sheet pdf
- Barney-s campfire sing along vhs vhs tape
- Autodesk inventor 2013 descargar
- Standard poster frame sizes
- Emcee send off wedding script
- Download full version of sql server 2012
- Direct3d overrider windows 10 download
- Best online webm to gif converter
- Loading gif aesthetic transparent
- Youtube music to mp3 download free
- Free small business software downloads
- Download putty and puttygen for windows 10
- Slime rancher multiplayer mod -
- Festo fluidsim 5 demo
- Free holiday border for word document
- Match baby animal names game
